No 6373
Wednesday 28 January 2015
Vol cxlv No 17
pp. 337–367
The Council begs leave to report to the University as follows:
1. In this Report the Council is seeking approval in principle for the construction of a new laboratory as set out below.
2. The strategy of both the Schools of the Biological Sciences and Clinical Medicine is to accommodate the following four complementary units, currently spread across multiple locations, into a single building to produce significant scientific synergies:
•The Cambridge Stem Cell Institute
•The Cambridge Institute of Therapeutic Immunology and Infectious Diseases (CITIID)
•The Cambridge Centre for Haematopoiesis and Haematological Malignancies (CCHHM)
•The Therapeutic Institute
The new laboratory will allow for recruitment to several Professorships and senior posts and encourage strategic collaboration as well as the efficient sharing of resources and equipment across 50–60 Research Groups representing strategic research priorities of both Schools.
3. Work in the new laboratory will include fundamental research as well as investigations into clinical and translational work, and biomedical applications. The research will address significant global issues including infectious disease and immunology, a wide range of haematological malignancies, and the potential of stem cell biology for generating, maintaining, and repairing tissues. The research has already received millions of pounds of funding from major grant-giving bodies and the new building will be designed to maximize the potential for world-leading research and collaboration.
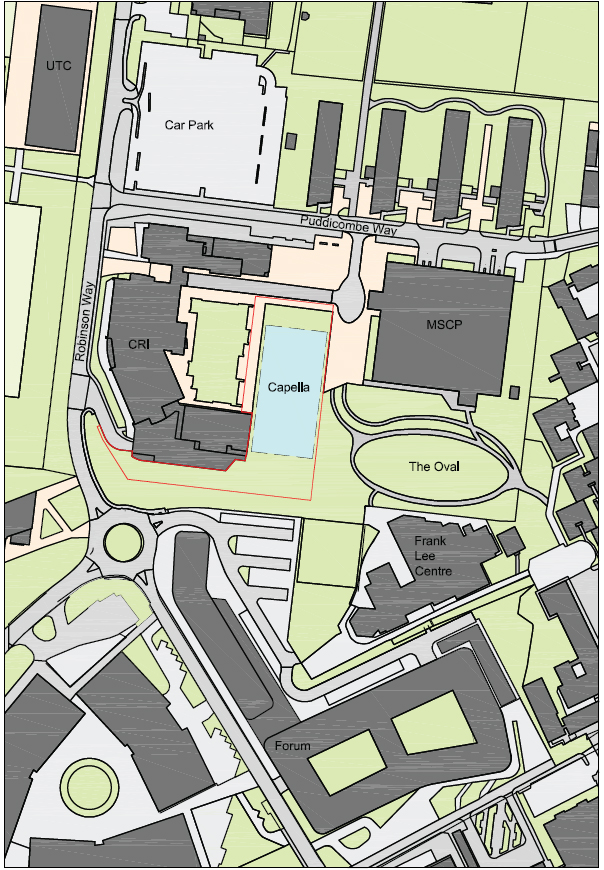
4. The laboratory is to be constructed on University land located within the Cambridge Biomedical Campus on Puddicombe Way between the Li Ka Shing Centre and the multi-storey car park. It will comprise a gross internal area of 18,000m2 of wet and dry laboratory space with shared core facilities. The design will focus on efficiency and environmental sustainability.
5. The total cost of the proposed development has been estimated at approximately £94m. A grant of £25m has been awarded to CITIID from HEFCE’s UK Research Partnership Infrastructure Fund (UKRPIF) 2015–16 and £40m is allocated within the Capital Fund. Philanthropic donations totalling £7m have been committed to CCHHM (£2m) and the Therapeutic Institute (£5m). The balance of funding is to be raised by the Schools of the Biological Sciences and Clinical Medicine, which have underwritten the project.
6. A concept paper for the project was approved by the Planning and Resources Committee on 22 May 2013. Further details relating to the design, maintenance, and recurrent costs, with proposals for funding, will be brought to future meetings of the Buildings Committee and the Planning and Resources Committee. A Second-stage Report will be published in due course to seek approval for implementation of the project.
7. A plan showing the location of the proposed building is shown below.
8. The Council recommends:
I. That approval in principle be given for the construction of a new building on Puddicombe Way for the Schools of the Biological Sciences and Clinical Medicine.
II. That the Director of Estate Strategy be authorized to apply for detailed planning approval in due course.
|
27 January 2015 |
L. K. Borysiewicz, Vice-Chancellor |
Helen Hoogewerf-McComb |
Shirley Pearce |
|
Ross Anderson |
Richard Jones |
John Shakeshaft |
|
|
Richard Anthony |
Fiona Karet |
Susan Smith |
|
|
Jeremy Caddick |
Stuart Laing |
Evianne Van Gijn |
|
|
Ruth Charles |
Rebecca Lingwood |
Sara Weller |
|
|
Anne Davis |
Susan Oosthuizen |
I. H. White |
|
|
David Good |
Rachael Padman |
A. D. Yates |
|
|
Nicholas Holmes |
The Council begs leave to report to the University as follows:
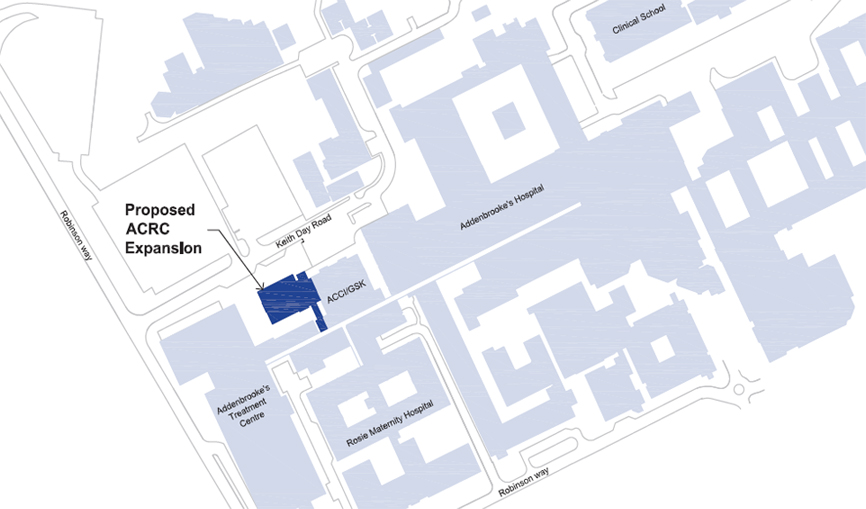
1. A First-stage Report on the expansion of the Addenbrooke’s Clinical Research Centre (ACRC) was submitted to the Regent House on 20 November 2013 (Reporter, 6326, 2013–14, p. 109) and approved by Grace 1 of 11 December 2013. This Second-stage Report is to update the Regent House on further development of the scheme and to seek approval for construction to proceed.
2. As stated in the First-stage Report, the ACRC is an indispensable component of research infrastructure on the Cambridge Biomedical Campus, making a significant contribution to translational research and generating high-impact publications. However, the ACRC is now operating at near full occupancy. In particular areas of the School of Clinical Medicine’s research (e.g. Metabolic Medicine and Cancer), there are pressing needs for additional space for research wards and clinics. There is no scope for physical expansion of the Clinical Research Facility or Clinical Investigation Ward within the existing envelope of the ACRC facility in the Addenbrooke’s Centre for Clinical Investigations/GSK building.
3. The new building will be constructed adjacent to the existing ACRC facility with a total additional floor space of 3,800m2 across five levels of clinical research accommodation, a rooftop plant area, and basement-level plant, service, and storage spaces.
4. In accordance with the Capital Projects Process, a Full Case was prepared by the School of Clinical Medicine and subsequently approved by the Planning and Resources Committee at their meeting on 25 June 2014.
5. The total cost of the building project has been estimated at £18.55m, of which £5.25m will come from the Wellcome Trust, £5m from the University’s Capital Fund, and £1m from the Evelyn Trust. The balance of funding is to be raised by the School of Clinical Medicine, which has underwritten the project.
6. Drawings of the proposed scheme are displayed for the information of the University in the Schools Arcade. A map showing the location is set out below.
7. The Council recommends:
I. That approval is confirmed for the expansion of the ACRC facility as proposed in this Report.
II. That the Pro-Vice-Chancellor (Planning and Resources) be authorized to accept a tender for the works, within the available funding, in due course.
|
27 January 2015 |
L. K. Borysiewicz, Vice-Chancellor |
Helen Hoogewerf-McComb |
Shirley Pearce |
|
Ross Anderson |
Richard Jones |
John Shakeshaft |
|
|
Richard Anthony |
Fiona Karet |
Susan Smith |
|
|
Jeremy Caddick |
Stuart Laing |
Evianne Van Gijn |
|
|
Ruth Charles |
Rebecca Lingwood |
Sara Weller |
|
|
Anne Davis |
Susan Oosthuizen |
I. H. White |
|
|
David Good |
Rachael Padman |
A. D. Yates |
|
|
Nicholas Holmes |